

The Protein Factory 2.0
Welcome to a world of new solutions

University of Insubria leads the SUST‑STAM project on stem cells and regenerative medicine
On 10 April 2026, the University of Insubria hosted in Varese the kick-off meeting of SUST‑STAM, a cross-border project focused on innovation in stem cells for regenerative medicine. Funded by the Interreg Italy–Switzerland Programme, the project aims to strengthen collaboration between universities, research centres, start-ups and companies operating in the life sciences sector.
As Italian lead partner, the University of Insubria contributes multidisciplinary expertise in biotechnology, engineering and economics, under the scientific supervision of Professor Loredano Pollegioni, Delegate for Research and Innovation. SUST‑STAM promotes a sustainable and competitive ecosystem for the development of advanced stem cell therapies, supporting research, technology transfer and industrial innovation across the Insubrian cross-border region.

Listen to an interview with Professor Elena Rosini, broadcast during the episode of Smart City on Radio 24 on January 19, 2026, titled "Lignina: come valorizzare il secondo più abbondante polimero in natura"
Approximately 30% of plant biomass is composed of lignin, the most abundant biopolymer in nature after cellulose, which provides rigidity to wood and plant tissues. It is one of the world’s most abundant renewable resources, yet also one of the least exploited due to its complex and variable chemical composition. Although it is a polymer made up of highly valuable compounds, at least 50 million tons of lignin are produced each year by the paper and cellulose industry and are used almost exclusively as fuel. In recent years, however, research has developed enzymes capable of breaking down lignin in a controlled manner and converting it into valuable chemical products. The technology is mature at the laboratory level and has significant potential to be economically sustainable. Now it is up to industry to take the next step.
Watch this interview that aired on December 22, 2025, on Sky Italia!
Interview with Loredano Pollegioni and Elena Rosini about the innovative biotechnological process that transforms PET from bottles into pure amino acids, thanks to engineered enzymes and microorganisms (ProPla project).
Listen to an interview with Professor Elena Rosini, broadcast during the episode of Smart City on Radio 24 on December 8, 2025, titled "La chimica al contrario: come smontare le molecole dei materiali tessili per poterli finalmente riciclare"
One of the most important frontiers in chemistry today is not about creating new compounds, but about dismantling old ones. The goal is to recycle materials at the molecular level: on one hand, to preserve the properties of materials as much as possible from one cycle to the next, minimizing degradation; on the other, to make it possible to recycle mixed or composite materials that cannot be separated. Among these are fabrics. The environmental impact of the fashion industry is one of the most significant, and fabrics are among the hardest materials to recycle, as most garments are made of mixed fibers that cannot be separated. This is where “molecular recycling” can make a difference.
OCTOBER 2025
ProPla project - new publication:
the enzymatic depolymerization of single-use plastic products, primarily made from PET, is now a reality. We report the development of an innovative tandem system process that uses engineered enzymes and Escherichia coli strains to depolymerize PET and convert the resulting products into enantiomers of alanine, useful amino acids, and starting point for further valorization processes.
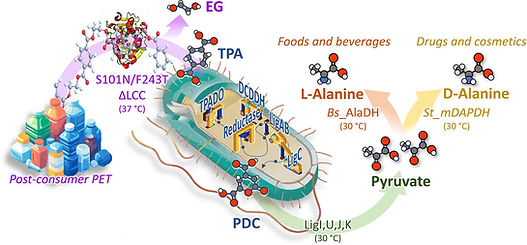
Rosini et al., 2025. Valuable compounds from pollutants: converting PET into enantiopure alanine. ACS Catal. doi: 10.1021/acscatal.5c06530.
Listen to an interview with Professor Loredano Pollegioni, broadcast during the episode of Smart City on Radio 24 on October 2, 2025, titled "Il batterio che trasforma la plastica in paracetamolo"
A team of scientists from the Wallace Lab at the University of Edinburgh has genetically reprogrammed Escherichia coli to convert a molecule known as terephthalic acid, derived from PET (the plastic used in bottles), into paracetamol (the active ingredient in Tachipirina).
The process is similar to fermentation, in which yeast transforms sugars into alcohol, and it occurs at room temperature with minimal environmental impact.
The idea, so far tested only at the laboratory level, represents another potential piece in the puzzle of the circular economy.
On April 2025 professor Elena Rosini has been elected as a member of the Board of Directors of Italian Biotechnologists for the years 2025-2027, in the role of Advisor for University Relations.
Italian Biotechnologists is the reference association for those working in the biotechnology sector in Italy.

On March 27-28, 2025, the final workshop of the second year of the ImmunoHub project, dedicated to "Immunotherapy: treatment and prevention of infectious and tumor diseases," was held at the University of Insubria.
The research program is coordinated by Professor Marco Terreni from the University of Pavia and involves a multidisciplinary team from Insubria led by Professor Loredano Pollegioni, the rector's delegate for Research and Technological Innovation. Immuno-Hub aims to develop a flexible national platform capable of providing rapid responses to new epidemiological needs through the design of biopharmaceuticals for immunotherapy. Researchers analyze both infectious diseases, such as SARS-CoV-2 and tuberculosis, and oncological pathologies, studying biomarkers for glioblastoma and gene expression in liver cancer and renal cell carcinoma.
The goal is to identify predictive markers of response to anti-tumor treatments and resistance to immunotherapy.
During the Workshop, Professors Gianluca Molla, Mauro Fasano, and research fellow Heather Bondi presented Bioinf-farma, a platform for predicting antigenic proteins for the development of innovative vaccines.
The event was also an opportunity to inaugurate the new CHO laboratory of the Department of Biotechnology and Life Sciences.
The project partners, in addition to the University of Insubria and the University of Pavia, include the University of Rome Tor Vergata, the IRCCS Foundation of the Policlinico San Matteo, the ASST Fatebenefratelli Sacco, the IRCCS Foundation of the National Cancer Institute, and the Scuola Normale Superiore of Pisa. The project is funded by the Ministero della Salute as part of the «Piano Operativo Salute - Traiettoria 4 – Interventi per la creazione di Hub delle Scienze della Vita nei settori della Farmaceutica, del Biomedicale e delle Biotecnologie».


In the photo, from left to right, are Prof. Flavia Marinelli, Director of the Department of Biotechnology and Life Sciences; Prof. Umberto Piarulli, Deputy Rector of the University of Insubria; Prof. Marco Terreni, coordinator of the Immuno-Hub project; Prof. Loredano Pollegioni, Rector’s Delegate for Research and Technological Innovation at the University of Insubria; Prof. Emilio Clementi, Director of the Clinical Pharmacology Unit of ASST Fatebenefratelli Sacco; Prof. Elena Rosini, head of the CHO laboratory; Dr. Natasa Kustrimovic, research fellow of the Immuno-Hub project; and Prof. Caterina Temporini, University of Pavia.

2025: Valentina Rabattoni has received funding from the Cariplo Foundation for the “SerMET” project, which investigates the role of serine in the human brain by studying the interaction between key enzymes in its biosynthesis.
In the photo, from left to right, are Professor Flavia Marinelli, Director of the Department of Biotechnology and Life Sciences, Dr. Chiara Tesi, Dr. Valentina Rabattoni, and Professor Loredano Pollegioni, the Rector’s Delegate for Research and Technological Innovation.
November 28, 2024: Presentation of the Rector’s Delegates at the University of Insubria
Professor Pollegioni has been entrusted by the new Rector of the University of Insubria with the responsibility for Research and Technological Innovation.
November 7, 2024: Study trip to Ecomondo: Biotechnology for Biobased and Health Industry students explore the circular bioeconomy
Students from the Master's degree program in Biotechnology for Biobased and Health Industry at the University of Insubria participated in a study trip to Ecomondo, the leading event for the green and circular economy, and a meeting point for industries, stakeholders, policymakers, opinion leaders, researchers, and institutions, held annually in Rimini.
Accompanied by the program director Gianluca Molla and professors Loredano Pollegioni, Elena Rosini, and Vincenzo Torretta, the students had the opportunity to learn about Italy's involvement in the circular bioeconomy. They were able to appreciate the diversity of approaches and numerous applications aimed at a more sustainable future.

21-24 August 2024: the 6th International Conference of D-Amino Acid Research (IDAR2024, Kanazawa, Japan)

MODULATION OF D-ASPARTATE CATABOLISM IN THE HUMAN BRAIN

ONE COIN, TWO SIDES: THE SERINOSOME

EFFECT OF HUMAN PHOSPHOSERINE PHOSPHATASE ASN133SER VARIANT ON SERINE SYNTHESIS: A CASE STUDY
Loredano Pollegioni, Valentina Rabattoni and Zoraide Motta are presenting their research findings at the 6th International Conference of D-Amino Acid Research in Japan.
15–19 July 2024: 21st International Symposium on Flavins and Flavoproteins (www.flavins2024.com / Atlanta, USA)

Prof. Loredano Pollegioni is presenting the intriguing story on "Human D-aspartate oxidase: a key player in D-aspartate brain metabolism" at the 21st International Flavins and Flavoproteins Symposium in Atlanta.
JULY 3, 2024: FEBS 2024 Closing Ceremony




JUNE 29 - JULY 3, 2024
The Protein Factory 2.0 lab members are presenting their research projects at the FEBS 2024 Congress (Milano MiCo Convention Centre).









JUNE 29, 2024
The young researchers from our laboratory presented their research projects to curious children and adults at the Città Studi Botanical Garden of the University of Milan.



JUNE 28, 2024



Nobel Prize winner for Medicine Bruce Beutler was our guest at the conference "Immunology and innovative therapies: from tumors to infections" held at the Salone Estense in Varese. Beutler presented his research project on the possibility of inducing tumor resistance in mice and, in the future, in humans. The conference was organized as a satellite event of the prestigious FEBS conference (Milan, June 29 - July 3) by Professor Loredano Pollegioni and the Department of Excellence in Economics at Insubria, headed by Professor Maria Cristina Pierro.
OCTOBER 2023
Professor Loredano Pollegioni has been awarded Ambassador of Milan for having supported Milan's candidacy as the host city of the FEBS 2024 Congress



Our PhD students Davide Miani and Filippo Molinari attended the post-graduate course in "Industrial biotechnology for lignocellulose based processes"


New publication:
the human flavoenzyme D-aspartate oxidase (hDASPO) controls the level of D-aspartate in the brain, a molecule acting as an agonist of NMDA receptors and modulator of AMPA and mGlu5 receptors. hDASPO-induced D-aspartate degradation prevents age-dependent deterioration of brain functions and is related to psychiatric disorders such as schizophrenia and autism.
Notwithstanding this crucial role, less is known about hDASPO regulation. This work represents the first study providing an insight into the regulation of hDASPO, the main enzyme controlling D-Asp availability in human tissues.
Rabattoni et al., 2023. On the regulation of human D-aspartate oxidase. Protein Sci. doi: 10.1002/pro.4802.

In U87 cells stably expressing pLG72-ECFP and transiently expressing hDASPO, PLA analysis showed that pLG72 (likely exposed on the outer membrane of the mitochondria) interacts with hDASPO.
Red: proximity spots indicating hDASPO-pLG72 interaction.
Green: cytoskeleton stained with Phalloidin CruzFluor™ 488.
Scale bar = 10 μm.
SEPTEMBER 2023
Inauguration of the new laboratory "Mirella Pilone"




The cover of the next FEBS Journal issue is related to our recent work on serine metabolism, in memory of Nadia Canu

Astrocytes are the major source of L-serine in the brain, which is synthesized from the glycolytic intermediate 3-phosphoglycerate through the phosphorylated pathway.
L-Serine is the precursor of the two main co-agonists of the N-methyl-D-aspartate receptor, glycine and D-serine. Here, we exploited a differentiation protocol, based on the generation of human mature astrocytes from neural stem cells, and investigated the modification of the proteomic and metabolomic profile during the differentiation process. The results underline how metabolism changes during astrocyte differentiation, highlight that D-serine synthesis is restricted in differentiated astrocytes and provide a valuable model for developing potential novel therapeutic approaches to address brain diseases, especially the ones related to serine metabolism alterations.
JUNE 2023
New instrument in our laboratory!
Opentrons® OT-2 liquid handler

MAY 2023
Our review on the enantiospecific interaction between proteins and chiral amino acids has been selected as Front Cover on Symmetry
Both L- and D-amino acids play key roles in the biochemical structure and metabolic processes of living organisms, from bacteria to mammals. In this review, we describe the molecular basis of the ability of some of the most important enzymes involved in the metabolism of amino acids to discriminate the opposite enantiomers. Our analysis showcases the power of natural evolution in shaping biological processes. Intriguingly, the active sites of enantioselective enzymes are frequently related by mirror symmetry.
MARCH 2023
Our review on the biological valorization of lignin components into value-added bioproducts has been selected as Front Cover on Catalysts
The biological valorization of lignin components into value-added bioproducts is expected to generate considerable market growth and to make the lignin valorization process profitable and sustainable in the near future. Recent advances in the field of systems metabolic engineering represent a comprehensive proof of concepts for great potentiality of renewable biomasses-derived aromatics.
Rosini E., Molinari F., Miani D., Pollegioni L. 2023.
Catalysts. https://doi.org/10.3390/catal13030555

FEBRUARY 2023

New publication:
this work proposed a setup of an efficient and green process for producing such valuable compounds from renewable feedstocks such as lignin and wheat bran.
Our catalytic system represents a sustainable and cost-competitive process to generate cis,cis-muconic acid, a building block for the synthesis of plastic materials.
The success of the proposed process makes our whole-cell system well suited to set up several innovative processes aimed at generating bioproducts and bioplastic monomers: bacterial lignin valorization seems no more a “field of dreams”.
JANUARY 2023
We produced a newly engineered biocatalyst that breaks down more than 99.2% of disposable post-consumer plastic waste in water and within hours (without adding chemicals or using high temperatures).
Watch for yourself!
Enzyme-based plastic degradation represents a valuable tool and a feasible solution for developing sustainable, environmental-friendly, and cost-effective plastic recycling processes from the circular economy perspective.
https://doi.org/10.1111/febs.16736.
Conclusion of the research project "Dissecting serine metabolism in the brain"
On 26 January 2023, professor Loredano Pollegioni organized an event to mark the conclusion of "Dissecting serine metabolism in the brain", a research project by The Protein Factory 2.0 (Università degli Studi dell'Insubria) in collaboration with Università degli Studi di Milano, Università degli Studi di Roma "Tor Vergata", Università degli Studi di Milano-Bicocca and Università degli Studi di Parma, funded by MIUR (PRIN 2017).
Watch the videos presenting the research results here!
A biochemical study:
An omics study:
Documentary on research at Insubria University by the journalist Alessandro Cecchi Paone
Professor Loredano Pollegioni presents the project carried out in collaboration with Dr. Elena Rosini and Professor Gianluca Molla on the generation of enzymes able to depolymerize lignin. This natural polymer can replace petroleum and allow the production of bioplastics.
New publication:
our optimized mammalian cells culture platform has been used to successfully express and purify a number of human recombinant proteins. As a paradigm, our established protocol has been efficiently adapted to rapidly produce the receptor binding domain (RBD) of SARS-CoV-2S protein in CHO cells.
Rosini and Pollegioni, 2022. Biotechnol. Appl. Biochem. https://doi.org/10.1002/bab.2409.


The research article by E. Vignali, et al. has been selected as front cover in ChemSusChem: this image shows the formation of low-molecular-weight compounds by the oxidative depolymerization of lignin by the laccase-Lig multienzymatic multistep system.
New publications:

In this work, we report a multi-omics analysis of the hippocampus of individuals with Alzheimer’s disease and healthy controls. Our study highlights a strong sex effect under normal aging and AD conditions: we observed a decrease in insulin response in AD when comparing the female with the male group; and also a significant modulation of serine metabolism.
A new multienzymatic multistep system for lignin valorization was designed and developed.
Lignin depolymerization through selective and specific enzymatic treatments is a key aspect in the biotechnological valorization of this biopolymer.
Qualitative and quantitative analyses permitted a more comprehensive elucidation of the effect of each biocatalyst used towards the development of a suitable method to obtain value-added end products from technical lignins of different botanical origin.

Can you believe that plastic can be fully degraded simply by an enzyme in water and within hours? Watch for yourself!
We produced a newly engineered biocatalyst capable of breaking down more than 95% of PET in only 48 hours of incubation at 60 °C, without adding chemicals or using high temperatures. Enzyme-based PET biodegradation represents a useful tool and a feasible solution for the development of sustainable, environmental-friendly, and cost-effective processes for plastic recycling in a perspective of the circular economy.
Pirillo et al., 2022. Int. J. Mol. Sci. doi: 10.3390/ijms23010264.
New publication:

New review available on D-amino acids as novel blood-based biomarkers.
Murtas and Pollegioni, 2021. Curr Med Chem. doi: 10.2174/0929867328666211125092438.
OCTOBER 2021
New review available on anticancer therapy based on oxidases that produce reactive oxygen species.
Rosini and Pollegioni, 2021. Biofactors. doi: 10.1002/biof.1789.
New publication:

in this work, we reported the first multi-enzymatic one-pot bioconversion of vanillin into cis,cis-muconic acid (ccMA), which is widely used for the production of biodegradable plastic materials. Our proposed biocatalytic system represents a sustainable alternative for the eco-friendly production of a high value-added compound from underutilized lignin, a primary target of the bioeconomy.
Professor Loredano Pollegioni has been elected Treasurer of the International Union of Biochemistry and Molecular Biology.



Antibody-drug conjugates, also named “armed antibodies”, can direct drugs to specific tissues. Here an evolved version of the ROS-generating enzyme D-amino acid oxidase was delivered to tumors by generating a chimeric protein with the F8 antibody against EDA of fibronectin, a protein abundantly expressed in the subendothelial extracellular matrix of tumor vessels. The recombinant chimeric protein produced in E. coli cells generates a strong cytotoxicity to tumor cells following the supplementation of the pro-drug D-alanine, especially when an inhibitor of the antioxidant systems is used.
Bioeconomy school: from basic science to a new economy. Lake Como School of Advanced Studies, 24-28 May 2021.
Organizing committee:
Loredano Pollegioni – Full Professor of Biochemistry, University of Insubria
Raffaello Seri – Full Professor of Econometrics, University of Insubria
Andrea Vezzulli – Associate Professor of Applied Economics, University of Insubria
Rendered with 3D Protein Imaging
New publication:
the human enzyme D-3-phosphoglycerate dehydrogenase catalyzes the first step of the L-serine biosynthesis, which plays an important role in numerous physiological functions. This enzyme is involved in cancer progression and several neurological disorders. Here is our new publication on PHGDH biochemical properties, part of the PRIN project “Dissecting serine metabolism in the brain”. https://www.mdpi.com/1422-0067/22/8/4231/htm
The cover art by Valentina Pirillo has been selected in The FEBS Journal Cover Competition 2021:
this image is an artist representation of a plastic-degrading enzyme (PET hydrolase) that is correlated with the recent Emerging Methods and Technologies article published in The FEBS Journal: ‘Analytical methods for the investigation of enzyme-catalyzed degradation of polyethylene terephthalate’ by Valentina Pirillo, Loredano Pollegioni and Gianluca Molla.


-
Not only regulation of D-amino acids. We propose a new function for human D-amino acid oxidase: DAAO plays an important role in the maintenance of the homeostasis of gut microbiota through the production of two antimicrobial peptides in the intestinal lumen. Murtas et al., 2021. Cell Mol Life Sci. doi: 10.1007/s00018-020-03755-w.
-
Serum D- and L-serine levels can represent a suitable, easy-to-detect and precocious biomarker for the diagnosis of the Alzheimer's disease and its progression. Piubelli et al., 2021. Transl Psych. doi.org/10.1038/s41398-021-01202-3.
NOVEMBER 2020
Elena Rosini produced the recombinant SARS-CoV-2 Spike (S) protein. The coronavirus spike protein plays a key role in the early steps of COVID-19 viral infection, mainly through the interaction with the human angiotensin-converting enzyme 2 (ACE2) present on the surface of alveolar epithelial cells. Both the full-length and a truncated form (containing only the receptor binding domain) of S-protein are available at "The Protein Factory 2.0" lab and can be used to develop new diagnostic tools.


.jpg)