The Protein Factory 2.0
Welcome to a world of new solutions
Conclusion of the research project "Dissecting serine metabolism in the brain"
On 26 January 2023, professor Loredano Pollegioni organized an event to mark the conclusion of "Dissecting serine metabolism in the brain", a research project by The Protein Factory 2.0 (Università degli Studi dell'Insubria) in collaboration with Università degli Studi di Milano, Università degli Studi di Roma "Tor Vergata", Università degli Studi di Milano-Bicocca and Università degli Studi di Parma, funded by MIUR (PRIN 2017).
Watch the videos presenting the research results here!
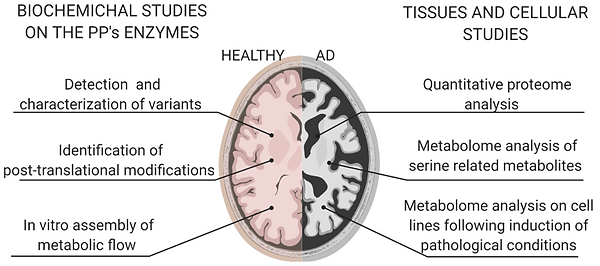
A biochemical study:
An omics study:
Dissecting serine metabolism in the brain
PI: Loredano Pollegioni
Participants:
Silvia Sacchi, Giulia Murtas, Valentina Rabattoni, Elena Zerbini, Zoraide Motta, Daniele Riva
Publications:
-
Murtas G, et al. 2020. L-serine synthesis via the phosphorylated pathway in humans. Cell Mol Life Sci. doi: 10.1007/s00018-020-03574-z.
-
Murtas G, et al. 2021. Biochemical and biophysical characterization of recombinant human 3-phosphoglycerate dehydrogenase. Int. J. Mol. Sci. https://doi.org/10.3390/ijms22084231.
-
Maffioli E, et al. 2022. Insulin and serine metabolism as sex-specific hallmarks of Alzheimer’s disease in the human hippocampus. Cell Reports https://doi.org/10.1016/j.celrep.2022.111271.
-
Rabattoni V, et al. 2023. The human phosphorylated pathway: a multienzyme metabolic assembly for L-serine biosynthesis. FEBS J. doi: 10.1111/febs.16787.
-
Riva D, Orlando M, Rabattoni V, Pollegioni L. On the quaternary structure of human D-3-phosphoglycerate dehydrogenase. Protein Sci. 2024, 33(8):e5089. doi: 10.1002/pro.5089.
Fundings:
PRIN 2017 (Grant 2017H4J3AS) to LP
Collaborations:
-
Gabriella Tedeschi (University of Milan, Milan, Italy)
-
Paola Coccetti (University of Milan-Bicocca, Milan, Italy)
-
Barbara Campanini (University of Parma, Parma, Italy)
-
Nadia Canu (University of Rome "Tor Vergata", Rome, Italy)

The cytosolic “phosphorylated pathway” for the de novo biosynthesis of L-serine (L-Ser) from the glycolytic intermediate 3-phosphoglycerate (3PG), is composed of three reactions catalysed by 3-phosphoglycerate dehydrogenase (PHGDH), phosphoserine aminotransferase (PSAT) and phosphoserine phosphatase (PSP).
In the brain, the “phosphorylated pathway” (PP) is the primary route for the synthesis of L-Serine (L-Ser), the precursor of glycine and D-Ser, two relevant coagonists of NMDA receptors, which are critically involved in synaptic plasticity, cognitive functions and cellular excitotoxicity. Either high or low levels of D-Ser are associated with NMDA receptors dysfunction and the related cognitive deficit that occurs in normal aging and Alzheimer’s disease (AD).
In this project, we plan to investigate the PP combining different biochemical approaches to perform analysis on cell model systems and brain samples aimed to: i) characterize the structural, functional and regulatory properties of the three involved enzymes (including splicing and point variants associated to pathological conditions); ii) perform extensive proteome and metabolome analyses.
The expected results, i.e. the system biology of serine circuit, might allow the identification of new mechanisms and targets for the treatment of pathologies in which the glutamatergic system is affected.